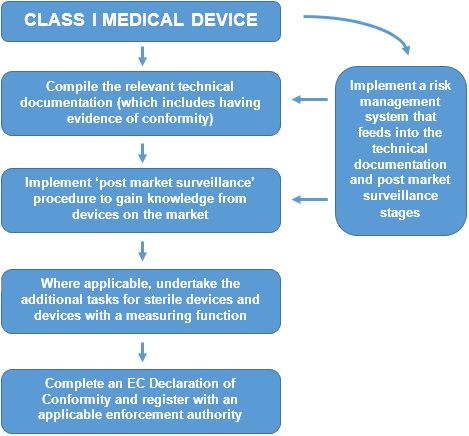
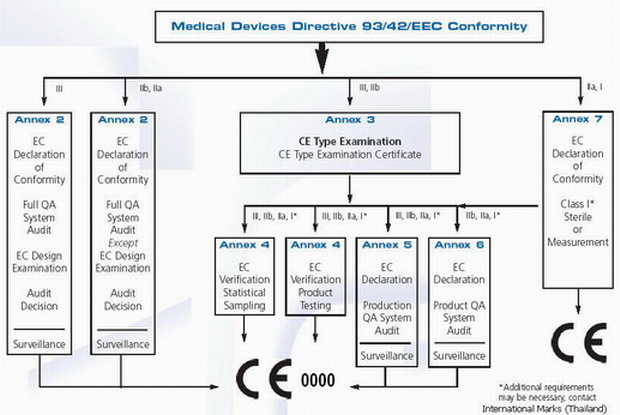
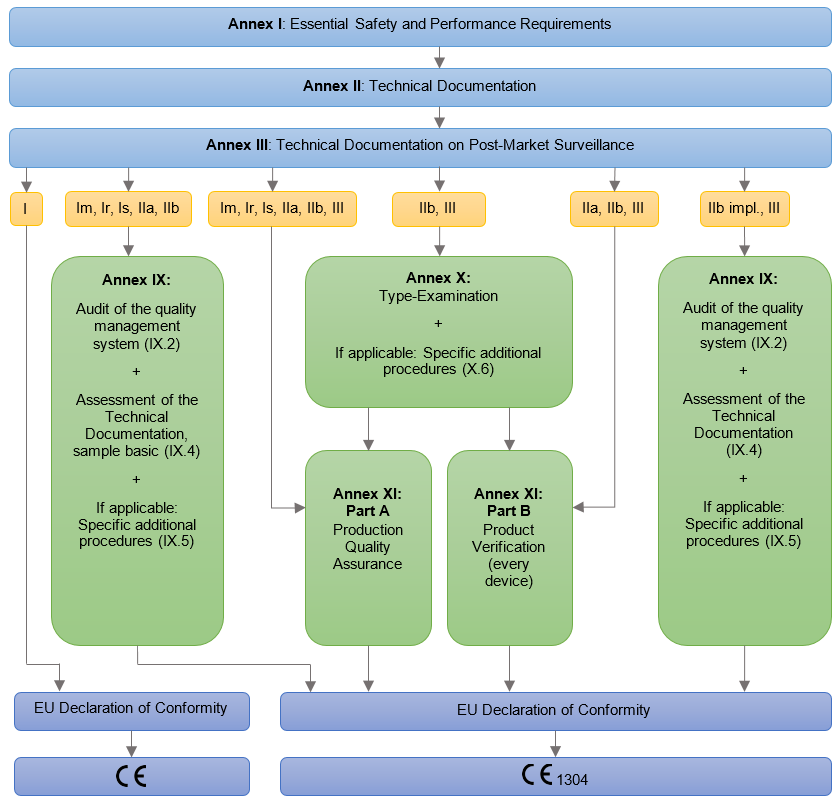
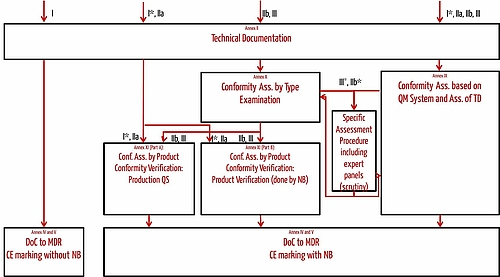
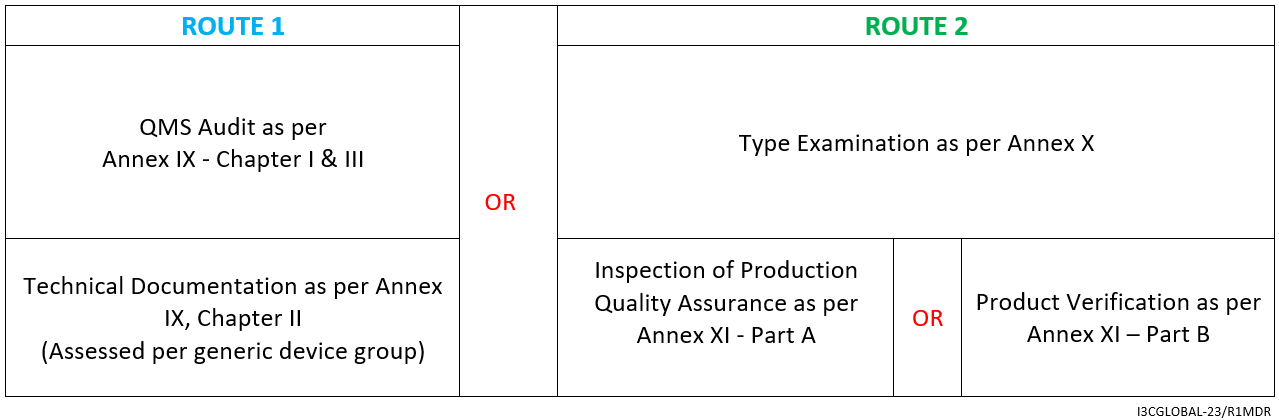
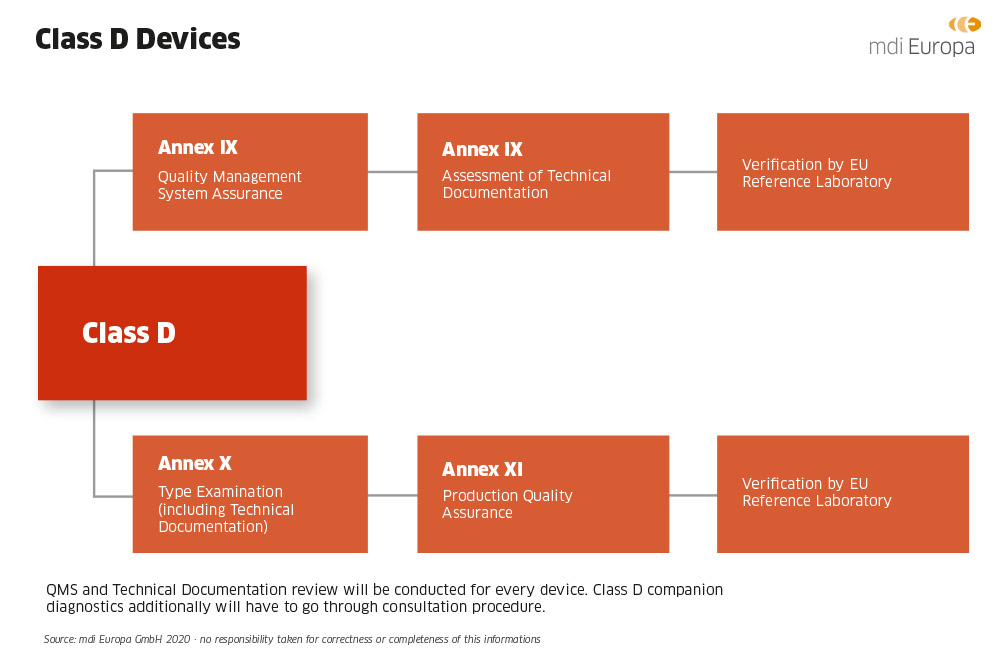
Medical Devices. Notified Bodies and the CE certification Process for Medical Devices. European Surgical Robotics Demonstration Day - PDF Free Download
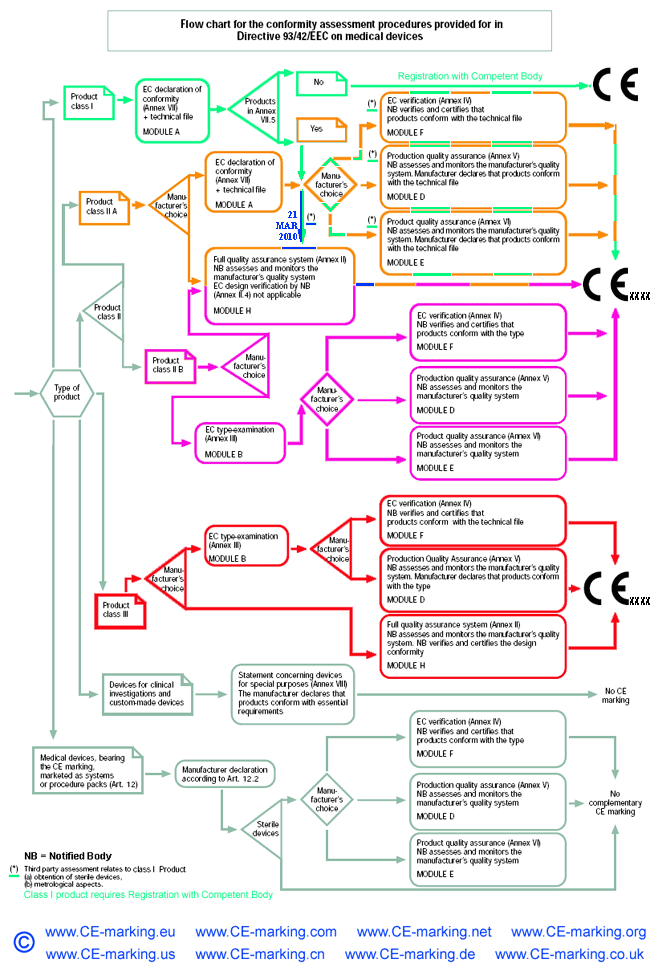
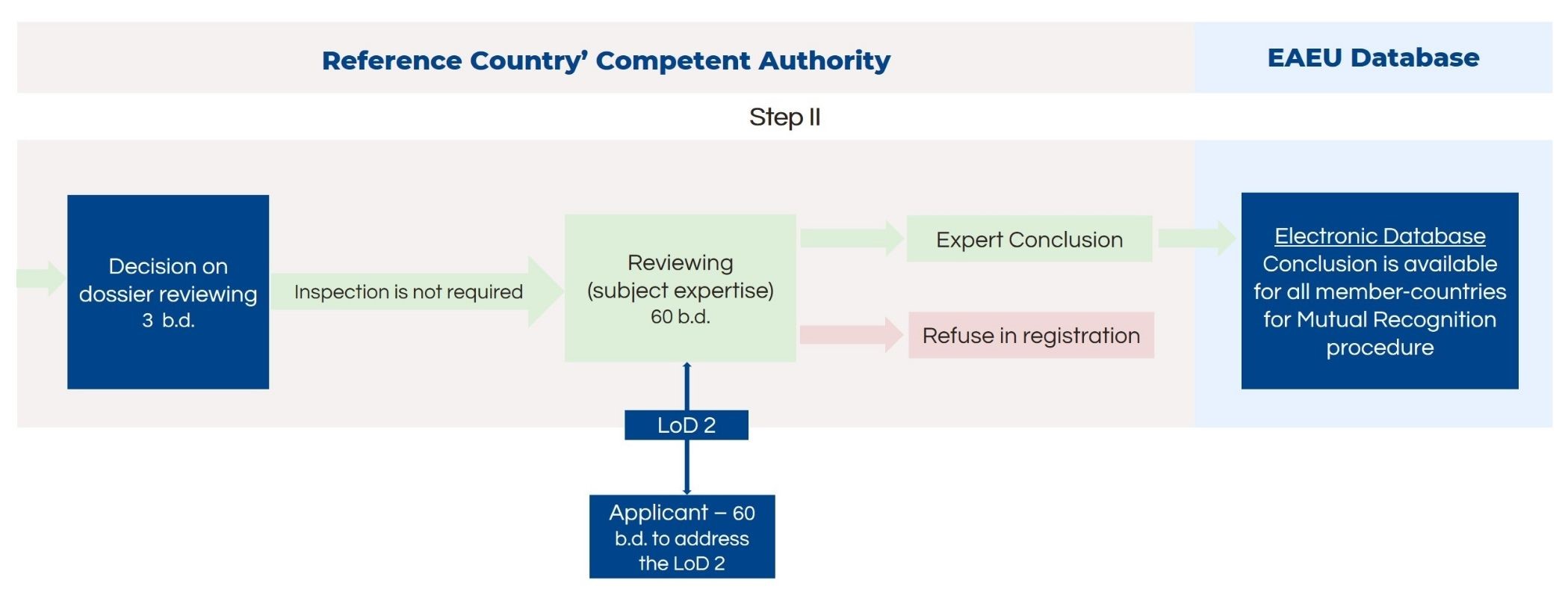
Guide on Class I (Is/Im) MDD- Medical Devices CE marking (mark) & European (EU) Authorized Representative service