
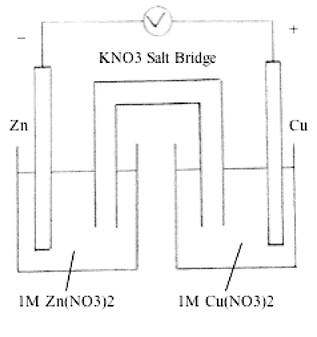
Zinc - Copper *Zn(s), Zn(NO3)2 (0.3 M) || Cu(s), CuCl2 (0.5 M) **1. Draw the schematic of the electrochemical cell that includes all the components (metals, solutions, salt bridges, voltmeters, etc. | Study.com

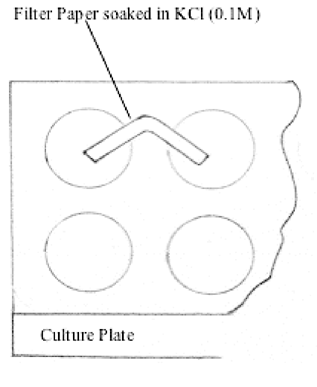
Filter Paper Salt Bridge in Potentiometric Titrations: An Undergraduate Laboratory Chemical Education Article

Filter Paper Salt Bridge in Potentiometric Titrations: An Undergraduate Laboratory Chemical Education Article
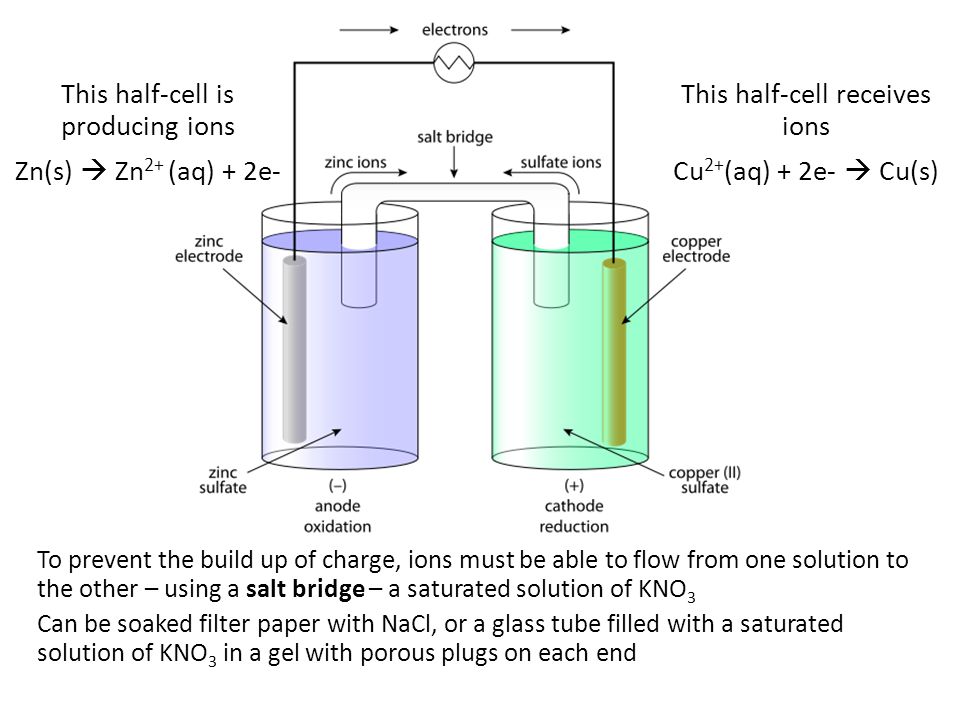

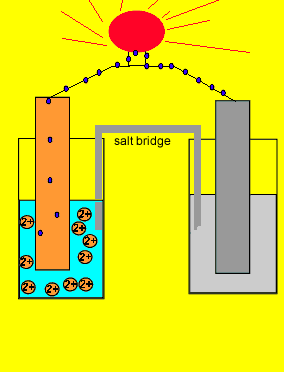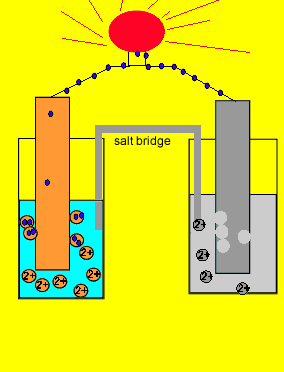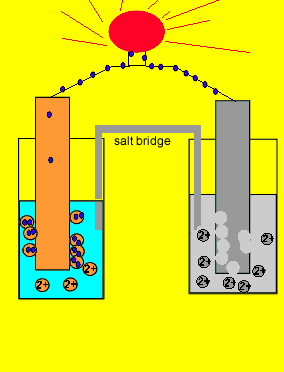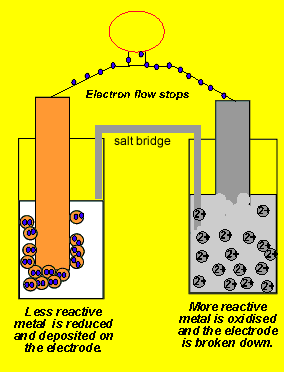

/saltbridge-5af43fcf875db900368d1853.jpg)


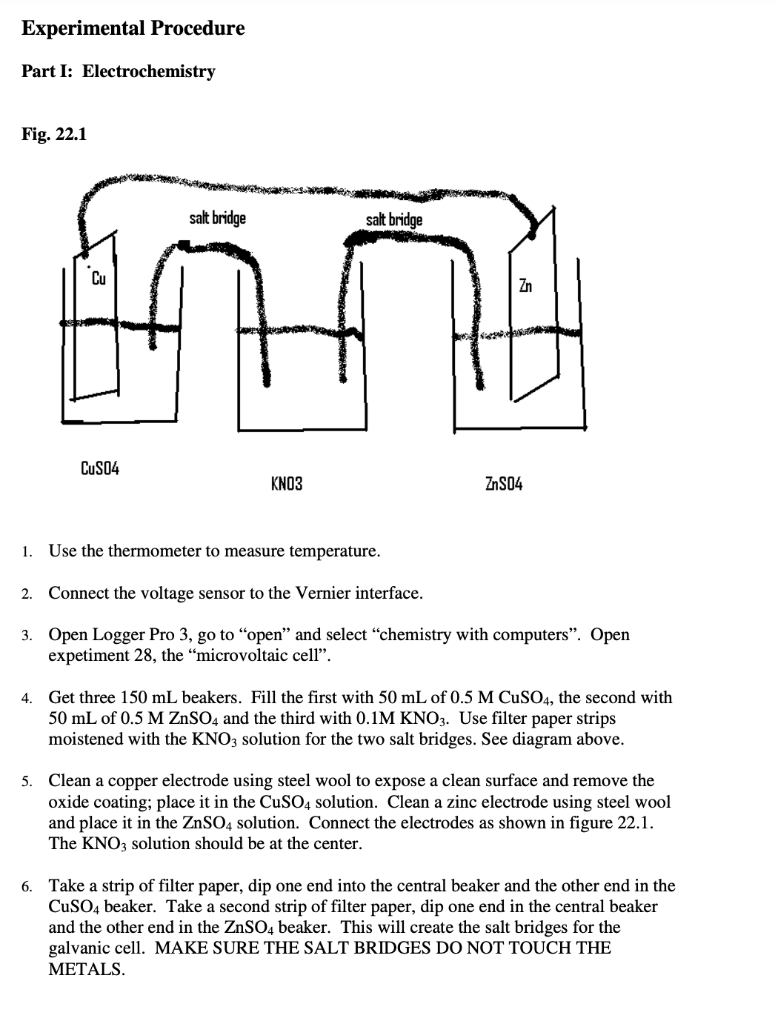


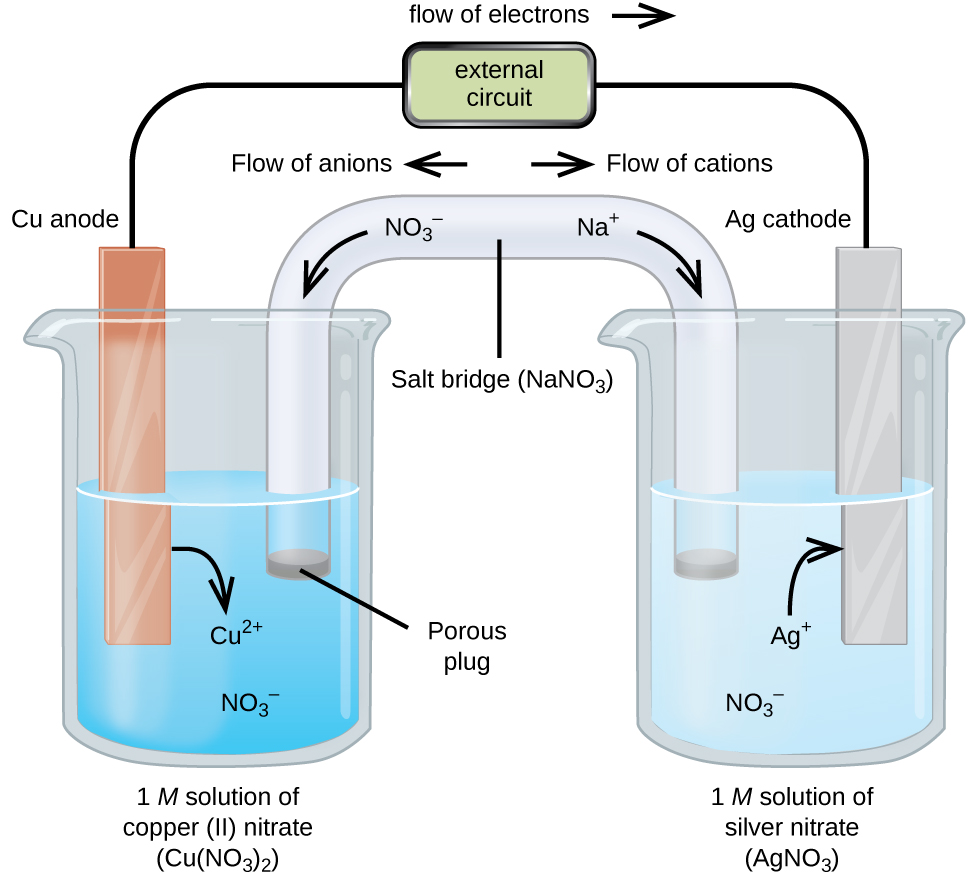


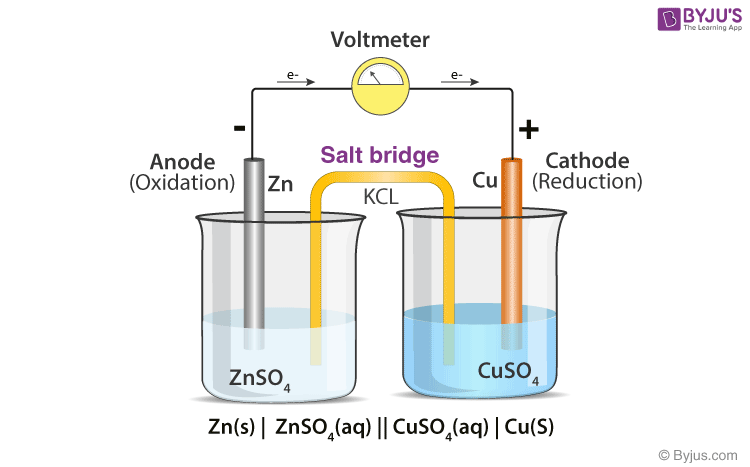
:max_bytes(150000):strip_icc()/GettyImages-10943484541-1145b5ddea574a1ab5d32aa398291feb.jpg)


![How well do force fields capture the strength of salt bridges in proteins? [PeerJ] How well do force fields capture the strength of salt bridges in proteins? [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2018/4967/1/fig-1-full.png)

/saltbridge-5af43fcf875db900368d1853.jpg)