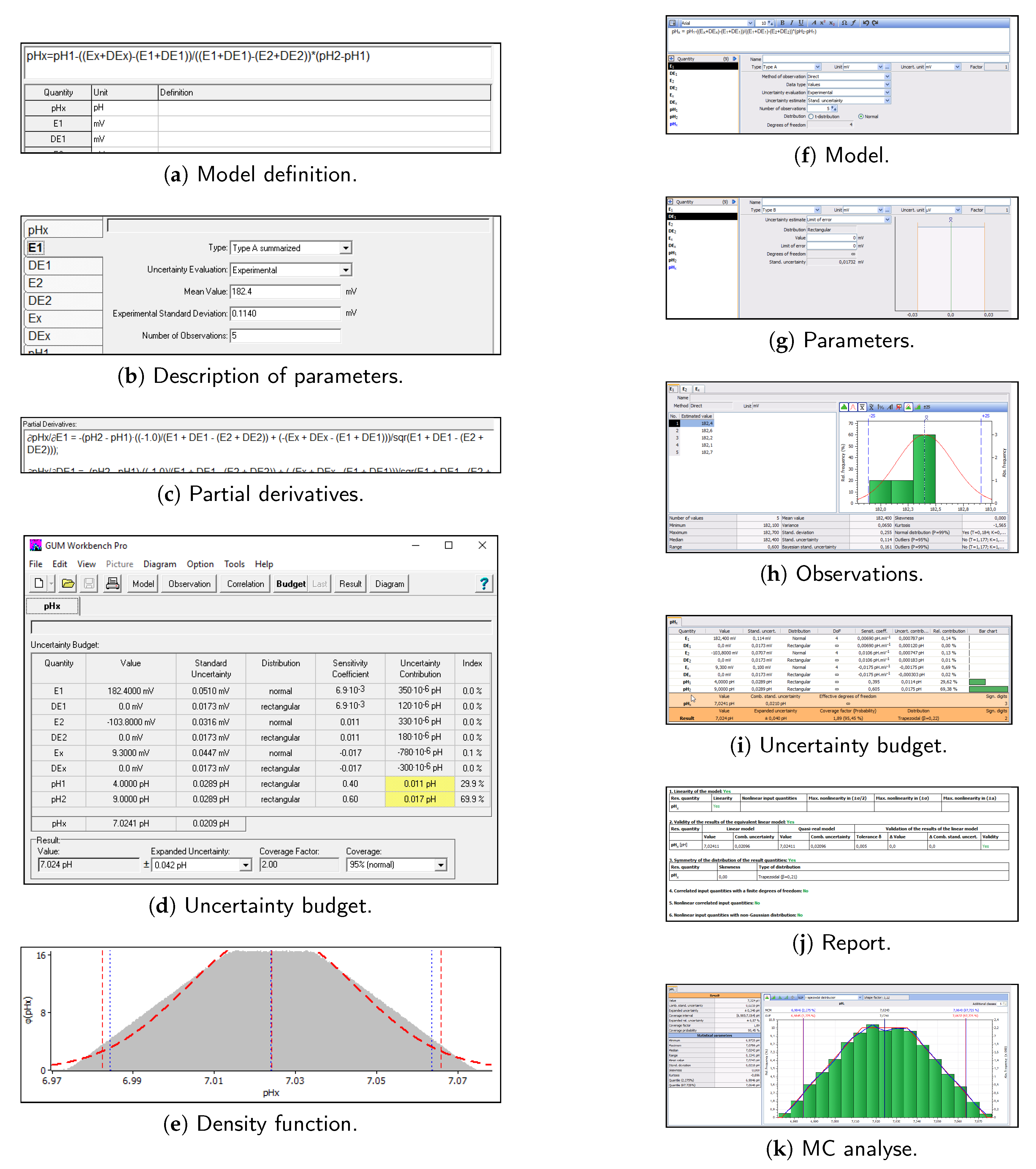
![PDF] Measurement Uncertainty Calculations for pH Value Obtained by an Ion-Selective Electrode | Semantic Scholar PDF] Measurement Uncertainty Calculations for pH Value Obtained by an Ion-Selective Electrode | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/225064b41ad8271042e31abea25e0eb460683afb/9-Table2-1.png)
PDF] Measurement Uncertainty Calculations for pH Value Obtained by an Ion-Selective Electrode | Semantic Scholar
![PDF] Measurement Uncertainty Calculations for pH Value Obtained by an Ion-Selective Electrode | Semantic Scholar PDF] Measurement Uncertainty Calculations for pH Value Obtained by an Ion-Selective Electrode | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/225064b41ad8271042e31abea25e0eb460683afb/12-Figure3-1.png)
PDF] Measurement Uncertainty Calculations for pH Value Obtained by an Ion-Selective Electrode | Semantic Scholar
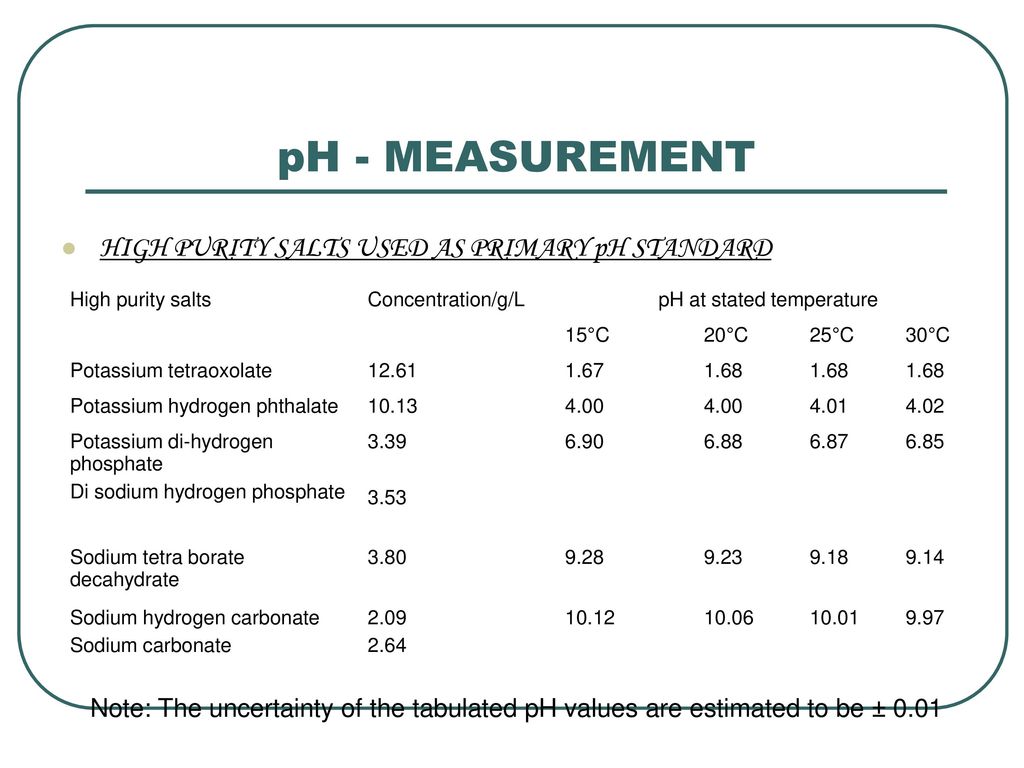
PH - MEASUREMENT pH: - It is a convenient measure of acidity / alkalinity of a aqueous solution at a specific temperature. It is measured on a continuous. - ppt download

PH - MEASUREMENT pH: - It is a convenient measure of acidity / alkalinity of a aqueous solution at a specific temperature. It is measured on a continuous. - ppt download

Combined Measurement Uncertainty for pH-Values Using Certified Reference Materials in Potentiometric Measurements with Glass Electrodes | SpringerLink

SOLVED: Select the sources of error associated with pH measurement with the glass electrode. (Select all that apply) junction potential drift, ambient brightness, uncertainty in solution volume, sodium or acid errors at